Carbonylation and Hydroformylation Reactions in Ionic Liquids
Hydroformylation reactions are important industrial reactions which involve the addition of CO and H2 molecules to unsaturated hydrocarbons to form aldehydes in presence of a homogeneous transition metal catalyst. Similarly, carbonylation reactions involve the addition of CO to hydrocarbons, creating a new C-C bond, typically with alcohols to give carboxylic acids.
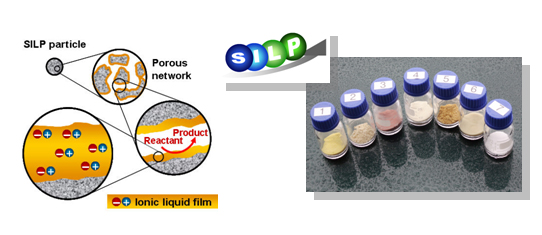
It is possible to carry out both hydroformylation and carbonylation reactions in ionic liquids with very active catalyst systems. Ionic liquids are ionic compounds that remain liquids at room temperature. Their physicochemical properties can be easily altered by modification of either the cation or anion, making the number of different ionic liquids potentially infinite. Unfortunately, the use of bulk ionic liquid systems needs lots of ionic liquid solvent, which still are relatively expensive compared to most common organic solvents. At the Centre for Catalysis and Sustainable Chemistry (CSC), we have developed a way to get around this by using the so-called Supported Ionic Liquid Phase (SILP) catalysts.. The SILP concept involves support of a thin layer of ionic liquid containing a catalyst on a porous support, which allows the required use of ionic liquid to be significantly reduced.
Projects at all levels are offered within Renewable Chemicals regarding gold-catalyzed conversion of renewable alcohols into carboxylic acids and derivatives as exemplified above. Depending on project direction and aim, projects will take place in cooperation with Danish companies.
For more information about the Centre of Catalysis and Sustainable Chemistry (CSC) and project opportunities contact:
Docent/Prof. Rasmus Fehrmann, building 206, room 116, Tel: 45252389, E-mail: rf@kemi.dtu.dk
Lektor/Assoc. Prof. Anders Riisager, building 206, room 125, Tel: 45252233, E-mail: ar@kemi.dtu.dk